Antitumor vaccine
The vaccine triggers mainly the cellular antitumoral immune responses which enable a complete destruction of different malignant and benign tumors.
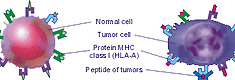
The vaccine possesses a vast and strong antitumoral properties due to a unique combination of telomerase antigen imitators together with more than 40 other tumor specific antigen imitators in its composition. The antigens, which are imitated by the drug, are present in cells of the most common malignant (see the list here) and benign tumors containing fibrotic and glandular tissues.
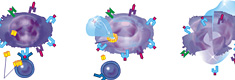
When the drug is introduced into an organism, a numerous clones of antitumoral lymphocytes are formed. In those cases when thus formed antitumor immunity could achieve a complete destruction of the tumors, relapses do not occur, due to the formation of the immunological memory.
The chances of the complete destruction of a tumor depend on:
1) Number of tumor cells (size of a tumor) and their mitotic activities;
2) Type of tumor- histological structures, antigen structures, the number of HLA-A class molecules on tumor cells;
3) Initial state of the immune system.
In the treatment of the malignant tumors it is most effective as an antimetastatic drug, when used in combination with surgical resection of a primary tumor which, depending on the stage of the cancers, results in to a complete destruction of tumor in 30-90% of cases. RESAN vaccine when used in the complex therapy of inoperable malignant tumor (T3-T4) can give a considerable therapeutic effect.
The drug does not render any toxic effect in normal cells of an organism, has no mutagenic and teratogenic effect and does not contain any cells and tissues of tumors. The main components of the vaccine are glycoproteins which imitate particular fragments of tumor antigens.
Latin name of the drug is RESAN. The antitumoral activities of the drug were researched and proved during 20 years. Preparation was registered in the public register of inventions; trademark registered 2009.03.19 No.29489, valid until 2026.04.28. Today, the irony in the cancer world is that in spite of the great need of such drugs, they are practically absent in the medical pharmaceutical markets.
![]()
A short instruction about the drug:
(full instructions are available with vaccine)
The presentation form:
Vials each with 200 mg of dry, white cryptocrystalline powder.
Composition:
Imitators of tumor antigens.
Pharmacological action:
The drug stimulates the formation of antitumoral lymphocytes and synthesis of cytokines, which augments the specific anti-tumor responses.
Indications:
• Usage of the drug as a vaccine for preventing malignant tumors in healthy people and people with positive heritable anamnesis and in those who have the tumor markers higher than in normal (elevated level of one or more of the following tumor markers: CA15-3, CA19-9, CA50, CA72-4, CA125, CA195, CA242, CA549, CA-SCC, CYRFA21-1, MUC-1, BCM, PSA, MCA, DUPAN-2).
• Use of the drug in immunotherapy of malignant tumors originated from connective, epithelial and glandular tissues mainly in combination with surgical treatments (T1-T2). For preventing relapses of malignant tumors after radical operation. Usage as antimetastatic drug in a combination with surgical resection of a primary tumor - destroys the metastases of size up to 10-50 cm3 (depending on the histological structure). In case of the inoperable malignant tumors (T3-T4) by using in a complex therapy (combine immuno-, chemo- and hormonotherapy).
• Use of the drug for the treatment of benign tumors, with general volume of 10-70 cm3, containing fibrotic (fibroma, fibroadenoma, fibromyoma) and glandular (adenoma, adenomatous polyps) tissues, as well as to prevent malignancy of benign tumors.
Gynaecological disorders:
1. Mastopathy (dishormonal hyperplasia of breast);
2. Endometrial hyperplasia;
3. Endometriosis;
4. Hysteromyomas (myoma of uterus).
Prostate pathology:
5. Benign Prostatic Hyperplasia;
6. Adenoma Prostate.
Thyroid pathology:
7. Thyroid Hyperplasia;
8. Hashimoto's thyroiditis.
The most appropriate time to inject the vaccine is 7-10 days prior the operation or 2-4 weeks post operation.
For women of childbearing age the vaccine should be administered during 7-21 days of their menstrual cycle.
Contraindications:
Absolute contraindications are absent. The drug is not recommended: during pregnancy; during 3-4 weeks after acute infectious diseases; during the activation of the chronic diseases; in case of allergic reaction to the drug during the previous administration.
Side effects:
The possible complications which may arise during our vaccination are similar to those in the case of other vaccinations in an organism. The general symptoms may be from mild cough, sneeze, urticaria, up to an attack of a bronchial asthma and anaphylactic shock. The general symptoms of the side effects may be due to the sensibilization of an organism by tumor antigens or introduction of a vaccine in the presence of an intercurrent infection. In such cases, the patients, within 7-10 days after vaccination, may feel a mild upset in their general health: weakness, nausea, sometimes joint pains.
In the patients suffering from an arterial hypertension, higher digits of arterial pressure can be registered within 1-3 weeks after vaccination.
All the patients should be under the doctor's observation during the administration of the drug. In case of allergic reactions, the further administration of the drug should be stopped, and should use antihistamine drugs.
Warnings:
To get the necessary immune answer, the level of leucocytes in peripheral blood ( in empty stomach) should be not less than 4.5·109 per liter and lymphocytes should be not less than 18%. If the patient, within last 3-6 months, had undergone chemotherapy, radiotherapy or took in immunosuppressors then the optimum immune answer may not be developed, as the immune system is hampered - needs 3-6 months to recover the immune system.
RESAN is not used for immunotherapy of cerebral tumors and neuraxis because the immunocompetent cells normally do not penetrate through the hematoencephalic barrier.
Mode of application:
The content of each vial is dissolved in 1.0 milliliter 0.9 % solution of sodium chloride (normal saline). The injections are carried out endermic, within 1 day, under a special scheme.
Duration of action:
The antitumor immunity is formed on 7-14 days after the vaccination and is maintained till the next revaccination period. The second vaccination is carried out with a single dose of 600-1000 mg (3-5 vials) of RESAN under a special scheme after 6 months. The first revaccination is carried out with a single dose of 400-800 mg after 1.5 years. The successive revaccinations are carried out in every 3-5 years.
Remarks:
The effectiveness of the immunotherapy is evaluated according to the degree of reduction in tumor marker levels or regression in clinical symptoms after 2, 6 and more months of vaccination. The overall effect is evaluated after second vaccination.
To increase the overall results of the cancer treatment in case of the inoperable malignant tumors (T3-T4) vaccine is recommended to administer in a complex therapy (combine immuno-, chemo- and hormonotherapy). If the blood level of interferon-gamma is low, it is reasonable to use interferon-gamma until the normalization of blood level of interferon-gamma. RESAN works well together with such chemotherapeutic drugs like Xeloda, Ftorafur, 5-ftoruracil and the other which have small toxicity to immune system (lymphopenia, leukopenia).
Storage, period of validity:
Store in dry original packings, in temperature between +15 - +25 0C. Period of validity is 2 years.
The drug has anticancerogenic effect against spontaneous, virus induced and cancerogenic-chemical induced malignant tumors. In case of the virus induced tumors it is necessary to carry out a preliminary effective anti viral therapy in order to have a successful anticancer vaccinotherapy.
Recently, our medical researchers have remarked that the vaccine administration in cancer patients leads to a stable and long term functional activation of CD56- positive lymphocytes, i.e. increase their cytotoxic activities and production of β-endorphins. As we know that the β-endorphins possess strong analgesic and sedative properties, the administration of vaccine releases pain considerably or stops progressing further in most of the cancer patients.
Thus administration of RESAN, the tumor-antigens simulator, enables:
- to prevent the formation of malignant tumors in many people, especially in those, whose relatives had suffered from oncologic diseases; i.e. genetically predisposed;
- to prevent malignancy (degeneration into malignant tumor) and to avoid operations in a number of benign tumor cases;
- to destroy small metastases and to prevent relapses after the surgical treatment of malignant tumors;
- to destroy the inoperable malignant tumors (T3-T4) by using it in a complex therapy;
- to improve the general health conditions and daily life-style, especially of those patients with highly aggressive or advanced cancers by releasing intensive pain symptoms which would avoid, in most of the cases, from taking massive analgesics.
This information is prepared mainly for the medical institutions and for the specialists working in the field of tumor immunology. Please have a close look at the proposal of possible mutual cooperation in the scientific research works. There is a possibility to get signed the contract papers officially for the delivery of the Imitators of the Tumor Antigens RESAN.
J. S. C. SCIENTIFIC RESEARCH ENTERPRISE RESAN
Street Tolstogo 1, Vitebsk 210026, Republic Belarus
Faxes:.+375-212-62-08-90;.+375-212-33-62-21;.+375-212-35-87-38.
Phones:..+375-296-30-03-90..
.E-mail:.resan@anticancer.net.
Exclusive Distributor of Vaccine RESAN in the Middle East:
MEDAWAR COMPANY S.A.L, Lebanon, Ain Saade Pine Garden;
medical consultant: Dr. Roukoz Medawar, Lebanon, Antelias, Diab Center 8 Floor
Phone: +9611686683; +9613293382;. Fax: +9611877676;
E-mail:.drromedawar@anticancer.net